Modernizing P&IDs for FDA-Regulated Manufacturing
Why Lifecycle Management of Engineering Drawings Is a Compliance Imperative
In a recent FDA inspection, an investigator cited the absence of as-built engineering diagrams and noted that existing air flow diagrams were “not current or up-dated.” The observation was not about a process failure. It was about drawings, the records that define a facility’s validated state.
Now imagine this scenario at your site. An FDA investigator reviewing your annual product review asks to see the change history for a CIP skid modification completed eighteen months ago. She wants the original P&ID and the revised version, along with the change request and qualification evidence. Your team has four hours before the next session. The engineering drawings are on a shared drive. The change control record is in the QMS. The validation protocol is in a binder. The SOP revision is in a document management system. Nothing connects.
In FDA-regulated manufacturing, P&IDs function as configuration records that define system boundaries and the as-qualified state of equipment. Lifecycle management means maintaining a controlled record across each configuration state, with revision history tied to approved change control and linked to qualification documentation.
Most P&IDs remain trapped in image-based PDFs. These files cannot be structurally searched or systematically connected to validation systems. The gap increases inspection risk by making it harder to demonstrate change control and documentation consistency when investigators examine how a validated state is maintained.
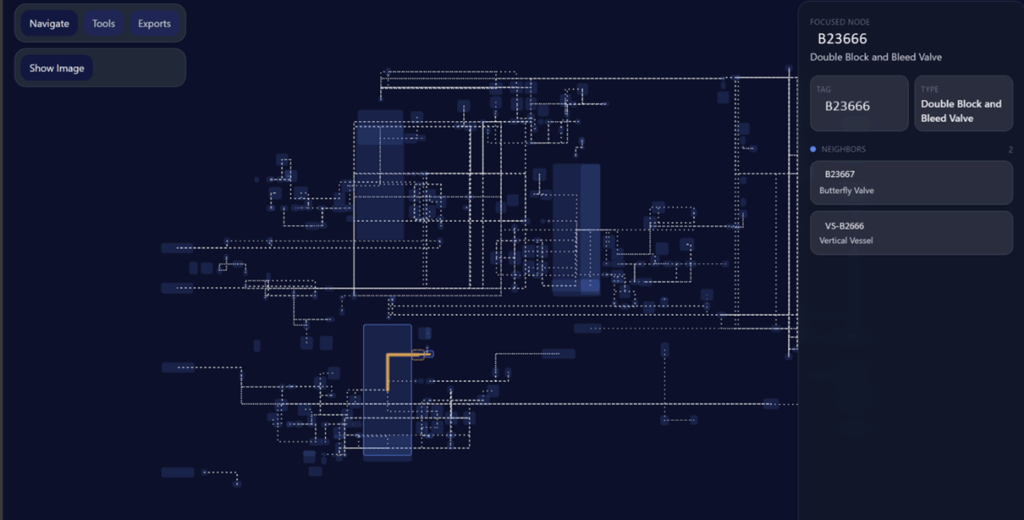
DeepIQ addresses this gap by converting static P&IDs into structured engineering datasets.
From Static Drawings to Controlled Configuration Records
FDA investigators expect engineering documentation to reflect the current validated state of a facility. When P&IDs exist only as image files, there is no reliable mechanism to determine whether a drawing reflects the as-built condition or when it was last reviewed.
DeepIQ converts scanned P&IDs into structured, machine-readable representations. Components and connections are captured as data within a digital model. The result is a governed engineering dataset that supports:
- Component-level visibility linking equipment tags to qualification records
- Comparison between P&ID revisions and referenced validation documentation
- Structured exports compatible with EAM or QMS platforms
- Facility-wide querying of engineering relationships
This approach moves P&IDs from static files to managed system representations.
Establishing a Defensible Revision History
A recurring theme in FDA enforcement actions is incomplete change history. During an inspection, the question is whether the organization can demonstrate when a system boundary changed and what documentation supported the release decision.
A lifecycle-managed P&ID allows teams to move from a drawing revision to the associated change record and supporting documentation.
Image-based P&IDs managed outside controlled systems typically lack structured metadata or reliable revision comparison. When revisions are overwritten, reconstruction of history becomes time consuming.
Structured P&IDs generated through DeepIQ create a chronological revision record. Each version of the engineering model can be preserved and compared, and associated with the change control record that authorized the modification. Rather than a generic revision label, the model can document that Valve V-204 material changed from SS304 to SS316 and record the approval date within the system.
Designed to Support GMP Documentation Expectations
In Good Manufacturing Practice environments, documentation must meet requirements for reliability and traceability. 21 CFR Part 11 establishes expectations for electronic record controls, while predicate rules define what documentation must exist.
Structured P&IDs support these expectations through revision tracking and user attribution within the engineering model. Platform-level access controls and audit logging can be configured to align with Part 11 principles when deployed within a validated environment.
Compared to unmanaged image files, structured P&IDs provide stronger support for:
- Computer-generated audit trails and role-based access controls
- Linkage between equipment elements and validation documentation
- Timestamped revision history that supports ALCOA+ principles
- Faster retrieval of engineering records during inspections
This shifts documentation management from reactive retrieval to structured oversight.
Strengthening Change Control and Validation
In regulated facilities, minor equipment changes can trigger broader documentation impact. A valve replacement on a WFI loop or an instrument adjustment on a clean steam system may affect multiple validation records.
When P&IDs are static images, assessing change scope is manual. Missed dependencies increase the risk of incomplete change documentation.
Because P&IDs are primary reference documents during Installation Qualification, discrepancies between executed work and referenced drawings can create deviations.
With structured P&IDs, teams can:
- Evaluate proposed changes within the context of the system model
- Generate object-level comparisons between revisions
- Link change orders to affected instruments within the engineering model
The result is more disciplined change evaluation and clearer visibility into system relationships.
AI-Assisted Engineering Queries
Once P&IDs are structured, they become queryable by AI systems operating on governed engineering data. Engineers and validation teams can ask:
- Which instruments control temperature in Reactor R-102?
- What systems connect to Pump P-14?
- List components within the CIP loop
Because the data is structured and connected, queries operate against the authoritative model. Outputs remain traceable to identifiable elements within the dataset.
The Result: A Digital Thread for Regulated Manufacturing
FDA-regulated manufacturers must maintain engineering documentation that supports both operations and compliance. Image-based P&IDs that are not part of controlled workflows make it more difficult to demonstrate change history and documentation alignment.
By modernizing P&IDs with DeepIQ, organizations convert static drawings into structured engineering records that support:
- Improved visibility into system relationships
- More consistent change impact evaluation
- Clear revision history across engineering states
- Documentation practices aligned with data integrity principles
- A foundation for AI-enabled engineering analysis
In an enforcement environment where FDA issued nearly 50 percent more CDER warning letters in FY 2025 and data integrity remains a frequently cited deficiency, the gap between static P&IDs and lifecycle-managed engineering assets is not simply a technology choice. It represents a measurable compliance risk for regulated manufacturers. DeepIQ converts static P&IDs into structured, governed engineering records that support the level of lifecycle control and traceability expected in FDA-regulated operations.